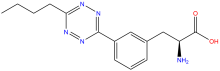
ncAA Structure (png, jpg, jpeg)
ncAA Foundational Publication Support
Jang, Hyo Sang, Subhashis Jana, Robert J Blizzard, Joseph C Meeuwsen, and Ryan A Mehl. (2020) 2020. “Access To Faster Eukaryotic Cell Labeling With Encoded Tetrazine Amino Acids.”. Journal Of The American Chemical Society 142 (16): 7245-7249. doi:10.1021/jacs.9b11520.
ncAA Utility
An amino acid that contains a 1,2,4,5-tetrazine-butyl group that can be used for bioorthogonal, click-chemistry reactions (bioconjugation) when reacted with strained trans-cyclooctenes (which can themselves be bound to various moieties such as PEG linkers, fluorophores, spin labels, etc.) either in vivo for eukaryotic systems or in vitro.
ncAA Source
Exogenous - Synthesized
ncAA Availability
Even though Tet3-butyl was synthesized in the attached paper (Access to Faster Eukaryotic Cell Labeling with Encoded Tetrazine Amino Acids), it can be purchased at the GCE4All site, here:
https://gce4all.oregonstate.edu/tetrazine-amino-acids
https://gce4all.oregonstate.edu/tetrazine-amino-acids
ncAA Synonyms
Tet3-Butyl
Tet-v3.0Bu
Tet3-bu
tetrazine3-butyl
tetrazine-v3.0 butyl
(S)-2-amino-3-(3-(6-butyl-1,2,4,5-tetrazin-3-yl)phenyl) propanoic acid
(2S)-2-amino-3-[3-(6-butyl-1,2,4,5-tetrazin-3-yl)phenyl]propanoic acid
Tet-v3.0Bu
Tet3-bu
tetrazine3-butyl
tetrazine-v3.0 butyl
(S)-2-amino-3-(3-(6-butyl-1,2,4,5-tetrazin-3-yl)phenyl) propanoic acid
(2S)-2-amino-3-[3-(6-butyl-1,2,4,5-tetrazin-3-yl)phenyl]propanoic acid
ChEBI ID
229638
PubChem Link