RS/tRNA Foundational Publication Support
Bryson, David I., Chenguang Fan, Li-Tao Guo, Corwin Miller, Dieter Söll, and David R. Liu. (dec) 2017. “Continuous Directed Evolution Of Aminoacyl-Trna Synthetases”. Nature Chemical Biology 13: 1253-1260. doi:10.1038/nchembio.2474.
RS/tRNA Pair Development Year
2017
ncAA(s) Incorporated
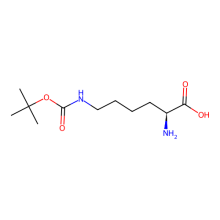
Nε-(tert-Butoxycarbonyl)-L-lysine (BocLys)
ncAA Structure (png, jpg, jpeg)
ncAA Utility
Extra-‐chromosomal array (low stability), biolistic bombardment. Scattered in different places in animal.
RS Organism of Origin
Parent RS
RS Mutations
V31I
T56P
H62Y
A100E
T56P
H62Y
A100E
tRNA Organism of Origin
Parent tRNA
tRNA Anticodon
CUA
RS/tRNA Availability
Addgene plasmid #104072
Used in what cell line?
RS/tRNA Additional Notes
Created to test if adding the "IPYE" mutations to various PylRSs would improve their activity. In BL21(DE3) cells, with 1 mM Boc-Lysine, produced 1.5 or 3-fold more protein for 3 different sfGFP constructs compared to the wild-type enzyme. The mutations are in the N-terminal tRNA-binding domain rather than the active site, and the increased catalytic performance is thought to arise from improved interactions with the tRNA. Although only the Boc-Lys ncAA substrate is listed here, it seems this mutant would in general be better to use than the wild-type enzyme to install any ncAA for which the wild type enzyme is useful.